Paper | 発表論文
2025
Z. Yang, K. Michigami, M. Ohashi
Chem. Lett. 2025, 54, upaf148.
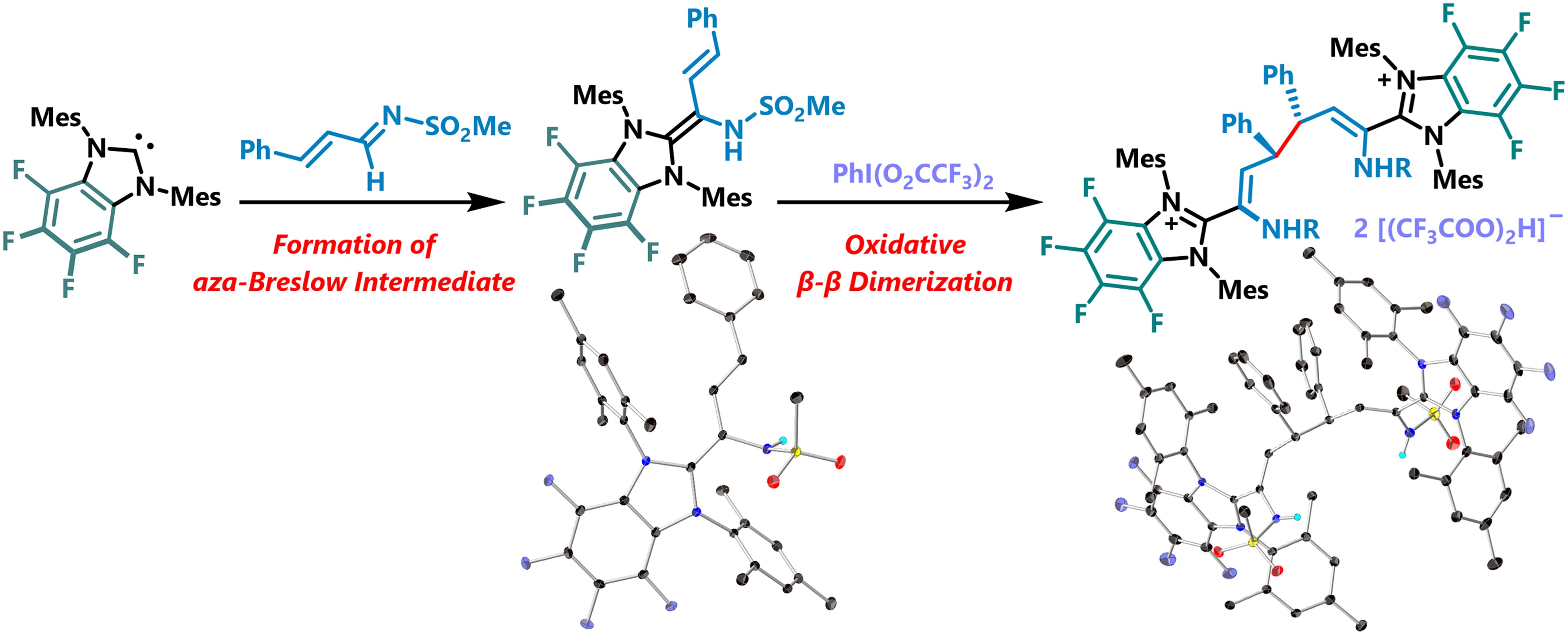
Abstract:
We herein disclose a unique oxidative β-β dimerization of conjugated N-sulfonylimines via umpolung reactivity mediated by fluorinated N-heterocyclic carbenes. The elusive key aza-Breslow intermediate was successfully isolated and characterized by single-crystal X-ray diffraction studies. This underwent net 1e– oxidation of aza-homoenolate upon treating with a hypervalent iodine reagent, and the successive tail-to-tail radical-coupling afforded the imidzolium-bound dicationic bis-enamide product. The comprehensive studies including single-crystal X-ray analysis elucidated the molecular structure and stereochemistry, revealing the formation of a sole diastereomer.
K. Michigami, D. Kawakami, I. Ueda, S. Ito, T. Ogaki, H. Ikeda, M. Ohashi
J. Mater. Chem. C 2025, 13, 1254-1262.
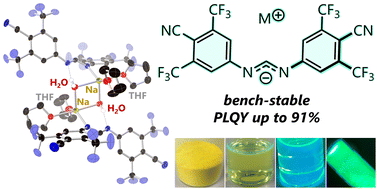
Abstract:
We developed the 4-cyano-3,5-bis(trifluoromethyl)phenyl group as a novel and potent monoaromatic acceptor unit. The CF3 and CN substitution effects were evaluated through the absorption and emission properties of readily accessible sodium N,N′-diarylformamidinate salts endowed with electron-withdrawing monoaromatic substituents on the nitrogen atoms. Among the salts examined, the amidinates substituted with 4-cyano-3,5-bis(trifluoromethyl)phenyl were remarkably stable at ambient temperature under air and room light, remaining unchanged for over a year.
2024
K. Michigami, Y. Otawa, Y. Kanda, D. Kawakami, M. Ohashi
ACS Catal. 2024, 14, 12647-12653.
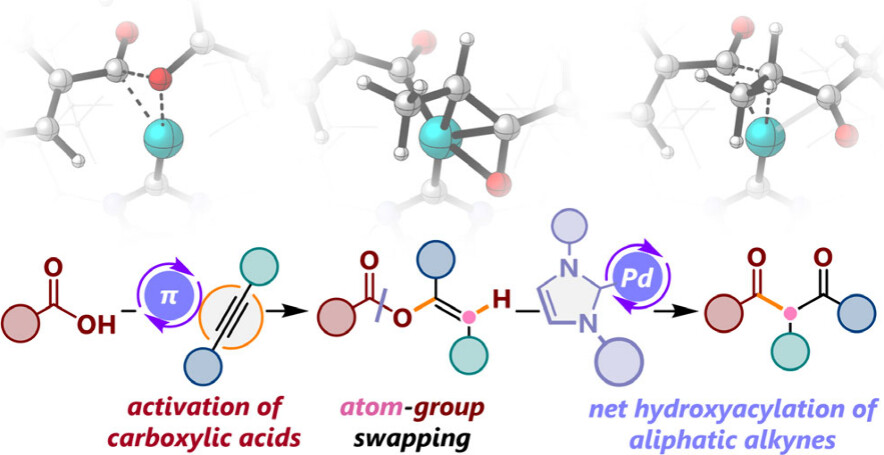
Abstract:
A catalytic system comprising Pd(0) and N-heterocyclic carbene (NHC) demonstrated remarkable efficiency in the acyl migration of enol carboxylates via the regioselective cleavage of the Cacyl−O bond. Employing NHC ligands with appropriate steric bulkiness and electron-donating ability facilitated the transformation of a diverse array of enol esters containing alkyl-substituted internal and terminal alkenes into unsymmetrical β-diketones.
Y. Miyazaki, K. Michigami, M. Ohashi
J. Am. Chem. Soc. 2024, 146, 8757-8767.
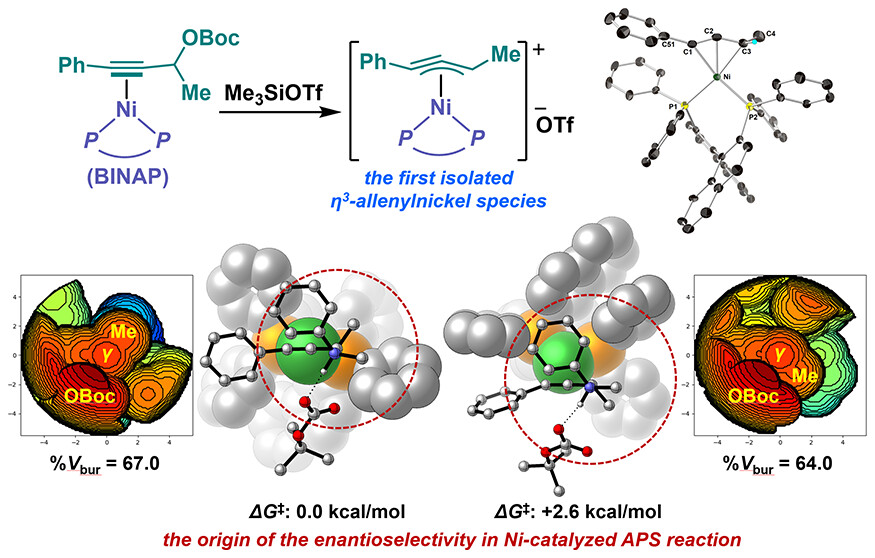
Abstract:
We succeeded in isolating the first example of a cationic η3-allenylnickel(II) complex via cleavage of the C−O bond in propargylic tert-butyl carbonate. Further stoichiometric and catalytic reactions employing the (R)-BINAP-coordinated cationic η3-allenylnickel(II) complex unequivocally demonstrated its involvement in the nickel-catalyzed APS reaction.
N. Taniguchi, M. Ueda, K. Michigami, S. Mori, M. Ohashi
Bull. Chem. Soc. Jpn. 2024, 97, uoae026.
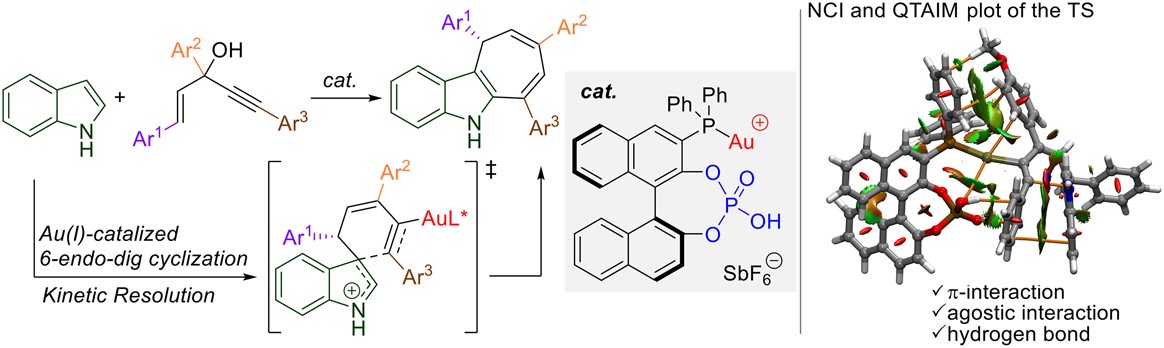
Abstract:
We designed and synthesized a novel gold(I) hybrid complex as a multifunctional catalyst that promotes asymmetric catalytic reactions through multipoint nonclassical noncovalent interactions in substrates that lack classical hydrogen-bond donors. We demonstrate its usefulness as a multifunctional catalyst by successfully developing the first catalytic asymmetric synthesis of dihydrocyclohepta[b]indoles.
2023
M. Ohashi, K. Ando, S. Murakami, K. Michigami, S. Ogoshi
J. Am. Chem. Soc. 2023, 145, 23098-23108.
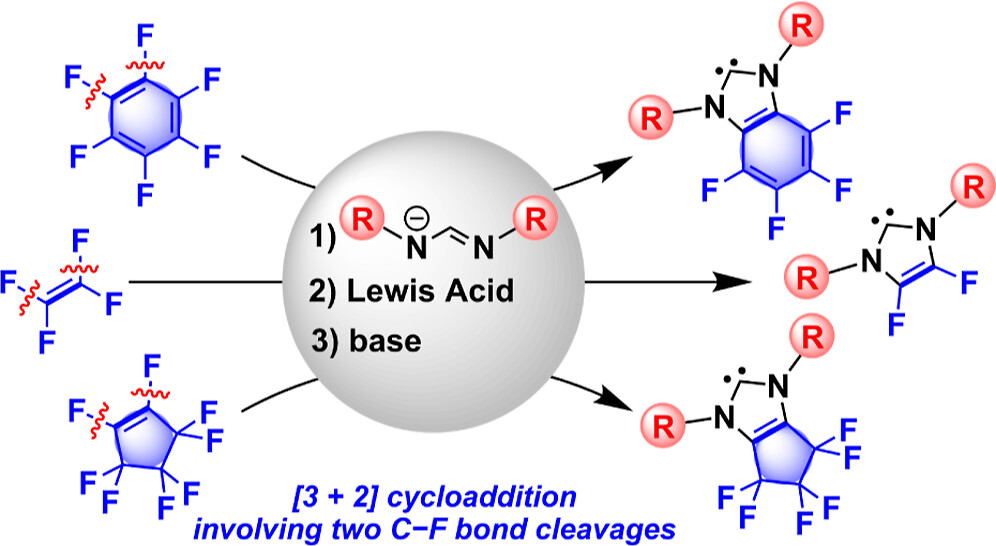
Abstract:
We developed a unique [3 + 2] cycloaddition reaction between formamidinates and perfluoroalkenes, such as tetrafluoroethylene and octafluorocyclopentene, in the presence of LiBF4 that affords 4,5-difluorinated imidazolium and perfluorocyclopentene-fused imidazolium salts. This protocol can also be applied to perfluorobenzene to furnish a tetrafluorobenzimidazolium salt.
Highlighted as a Cover Picture
2021
D. E. Sunagawa, N. Ishida, H. Iwamoto, M. Ohashi, C. Fruit, S. Ogoshi
J. Org. Chem. 2021, 86, 6015-6024.
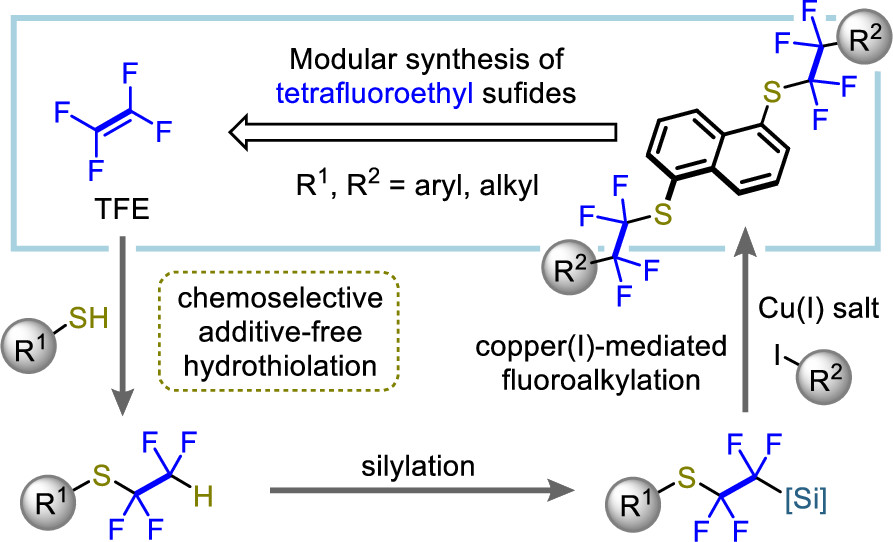
Abstract:
A modular synthetic method, involving a hydrothiolation, silylation, and fluoroalkylation, for the construction of highly functionalized fluoroalkyl sulfides has been developed. The use of the aprotic polar solvent N,N-dimethylacetamide is the key to success in the additive-free chemoselective hydrothiolation of tetrafluoroethylene, trifluorochloroethylene, and hexafluoropropene with various thiols.
N. Ishida, T. Adachi, H. Iwamoto, M. Ohashi, S. Ogoshi
Chem. Lett. 2021, 50, 442-444.
Abstract:
A novel copper-mediated N-fluoroalkylation of heteroarenes has been developed. Treating an in-situ-generated N–Cu complex with tetrafluoroethylene in the presence of iodoarenes furnished the corresponding N- and aryl-difunctionalized tetrafluoroethanes in up to 98% yield. This method enables the synthesis of tetrafluoroethylene-bridged heteroarenes in a one-pot reaction.
N. Ishida, H. Iwamoto, D. E. Sunagawa, M. Ohashi, S. Ogoshi
Synthesis 2021, 53, 3137-3143.
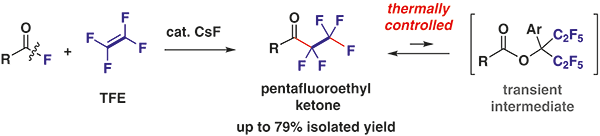
Abstract:
A catalytic method for the preparation of pentafluoroethyl ketones has been developed. The cesium fluoride catalyst can be used to convert acyl fluorides into the pentafluoroethyl ketones under tetrafluoroethylene pressure without generating stoichiometric quantities of chemical waste.
2020
Cleavage of C(sp3)–F Bonds in Trifluoromethylarenes Using a Bis(NHC)nickel(0) Complex
H. Iwamoto, H. Imiya, M. Ohashi, S. Ogoshi
J. Am. Chem. Soc. 2020, 142, 19360-19367.
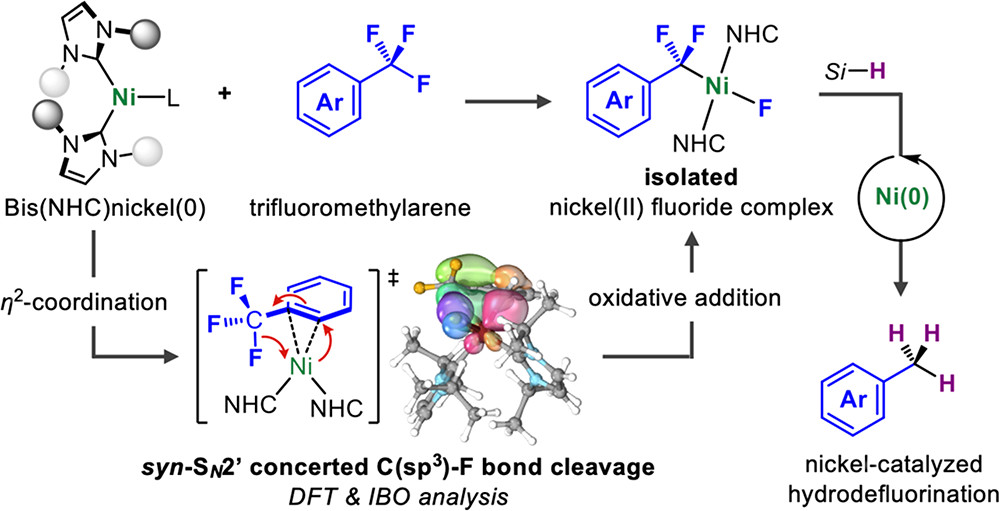
Abstract:
The oxidative addition of a single C(sp3)–F bond in trifluoromethylarenes to a transition-metal complex is described. A nickel(0) complex that bears two NHC ligands is able to cleave the C(sp3)–F bonds. Isolation and characterization studies suggested that the cleavage of the C(sp3)−F bond proceeds via a η2-arene nickel(0) complex.
Direct Transformation of Tetrafluoroethylene to Trifluorovinylzinc via sp2 C–F Bond Activation
K. Kikushima, Y. Etou, R. Kamura, I. Takeda, H. Ito, M. Ohashi, S. Ogoshi
Org. Lett. 2020, 22, 8167-8172.
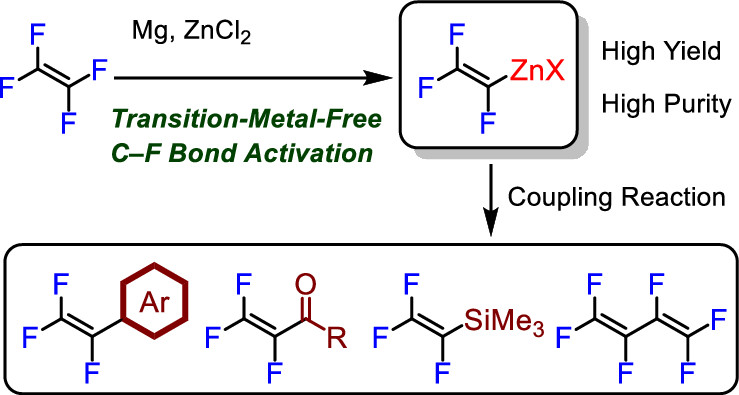
Abstract:
A novel synthetic approach for the preparation of trifluorovinylzinc chloride via a C–F bond activation of tetrafluoroethylene (TFE) is disclosed. The present system provides a practical synthetic route to various trifluorovinyl derivatives with very low energy consumption.
2019
H. Shirataki, T. Ono, M. Ohashi, and S. Ogoshi
Org. Lett. 2019, 21, 851-856.
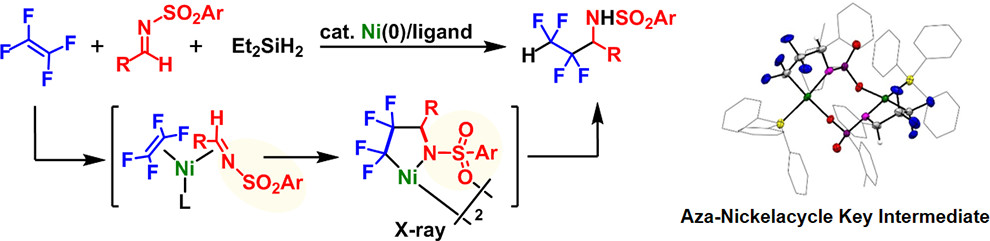
Abstract:
The nickel-catalyzed synthesis of a variety of fluorine-containing silyl ethers from tetrafluoroethylene (TFE) and aldehydes with silanes in a selective manner is disclosed. Stoichiometric reactions revealed that the oxa-nickelacycle, which is generated upon oxidative cyclization of TFE and an aldehyde with Ni(0), is the key intermediate, whose molecular structure was confirmed by single-crystal X-ray diffraction analysis.
Highlighted as a Cover Picture