Nanovaccine System for Cancer Immunotherapy
Immunity is a mechanism that protects the body from infections such as viruses and bacteria. Cancer immunotherapy is the application of immunity, which is intrinsic body's defense mechanism, to the treatment of cancer. If cancer immunotherapy can be established, it is expected to become the highly safe and personalized treatment.
Target-specific immunity induction occurs when antigen-presenting cells, mainly dendritic cells, present antigens for identifying abnormal cells to immunocompetent cells. Antigen presentation by antigen-presenting cells involves two types of molecules: MHC class I or class II. When an antigen is taken up by an antigen-presenting cell through phagocytosis, it is processed into peptide fragments in intracellular endosomes/lysosomes and presented on MHC class II molecules on the cell. Antigens presented on MHC class II molecules are recognized by helper T cells, leading to the induction of humoral immunity. When antigens are delivered in the cytosol of antigen-presenting cells, they are processed by proteasomes into peptide fragments, which are then presented on MHC class I molecules on the cells. Antigens presented on MHC class I molecules activate cytotoxic T cells, leading to the induction of cellular immunity.
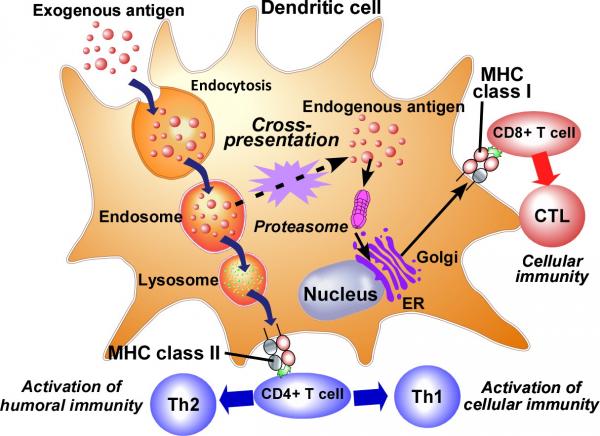
In cancer immunotherapy, cellular immunity plays an important role in eliminating abnormal cells such as cancer cells. Therefore, to realize cancer immunotherapy, it is essential to develop an antigen delivery system that delivers foreign antigens to the cytosol of antigen-presenting cells and induces cellular immunity through antigen presentation via the MHC class I pathway. We are conducting research on nanovaccine systems that use functional liposomes to deliver antigens to appropriate locations within cells and effectively induce immunity. We are also working to improve the performance of liposomes by incorporating various functional molecules that activate cancer immunity into them.
Papers on this theme
- E. Yuba, R. K. Gupta, Preparation of glycopeptide-modified pH-sensitive liposomes for promoting antigen cross-presentation and induction of antigen-specific cellular immunity, Biomater. Sci., 12, 1490-1501 (2024).
- E. Yuba, Y. Kado, N. Kasho, A. Harada, Cationic lipid potentiated the adjuvanticity of polysaccharide derivative-modified liposome vaccines, J. Control. Release, 362, 767-776 (2023).
- S. Yanagihara, N. Kasho, K. Sasaki, N. Shironaka, Y. Kitayama, E. Yuba, A. Harada, "pH-Sensitive branched β-glucan-modified liposomes for activation of antigen presenting cells and induction of antitumor immunity", J. Mater. Chem. B, 9, 7713-7724 (2021). DOI: 10.1039/d1tb00786f
- E. Yuba, Y. Sugahara, Y. Yoshizaki, T. Shimizu, M. Kasai, K. Udaka, K. Kono, “Carboxylated polyamidoamine dendron-bearing lipid-based assemblies for precise control of intracellular fate of cargo and induction of antigen-specific immune responses”, Biomaterials Science, 9, 3076-3089 (2021). DOI:10.1039/D0BM01813A
- E. Yuba, A. Yamaguchi, Y. Yoshizaki, A. Harada, K. Kono, “Bioactive polysaccharide-based pH-sensitive polymers for cytoplasmic delivery of antigen and activation of antigen-specific immunity”, Biomaterials, 120, 32-45 (2017). DOI: 10.1016/j.biomaterials.2016.12.021.
- E. Yuba, N. Tajima, Y. Yoshizaki, A. Harada, H. Hayashi, K. Kono, "Dextran derivative-based pH-sensitive liposomes for cancer immunotherapy", Biomaterials, 35, 3091-3101 (2014). DOI:10.1016/j.biomaterials.2013.12.024
- E. Yuba, A. Harada, Y. Sakanishi, S. Watarai, K. Kono, "A liposome-based antigen delivery system using pH-sensitive fusogenic polymers for cancer immunotherapy", Biomaterials, 34, 3042-3052 (2013).