Study Group 1 Elucidating the pathophysiological mechanisms of autoimmune skin diseases to develop novel treatments
Research
Study Group 1 Elucidating the pathophysiological mechanisms of autoimmune skin diseases to develop novel treatments
Research themeElucidating the pathophysiological mechanisms of autoimmune skin diseases to develop novel treatments
PI:Sho Hiroyasu
The immune system in human body distinguishes the "non-self" and eliminates it from the body to protect one's "self" from intruders. However, if it mistakenly recognizes parts of one's "self" as "non-self", the immune system attacks its own tissues, triggering various symptoms to cause "autoimmune diseases". There are multiple autoimmune disorders in dermatology field, many of them difficult to treat and whose treatments often have significant adverse effects. Therefore, safe and effective therapies are currently unmet clinical need in these refractory autoimmune skin diseases.
Our group is conducting research on various autoimmune dermatologic disorders, such as pemphigoid diseases and vitiligo. Through elucidating the pathophysiological mechanisms of these diseases, we aim to advance the dermatology field in such a way that benefits human health with development of novel treatment modalities, and further contribute to the development of skin science with deepening the understanding of the complex systems in skin immunity.
-
Pathological functions of pemphigoid-IgG
The pemphigoid diseases are a subgroup of autoimmune blistering disorders characterized by widespread subepidermal blisters on the body, often affecting elderlies. The current standard therapy is topical or systemic corticosteroid administration, which non-specifically targets the inflammatory response. Although application of corticosteroids is highly effective for most cases, which may be insufficient and are often associated with significant adverse effects. As such, safer and more effective treatments for this disease group are highly required in the clinical field. Since the number of patients is escalating because of increasing prevalence of risk factors such as aging, neurological conditions, and exposure to triggering drugs, requirement of the novel treatment options is also in rise.
Initial investigations into the pathogenic mechanisms of the pemphigoid diseases have established the presence of autoantibodies binding to antigens in the dermal-epidermal junction (DEJ). After these autoantibodies bind and deposit to the DEJ, complement activation, granulocyte recruitment, and release of various proteases result in the degradation of the adhesion proteins, leading to blister formation (Figure 1). In addition to this mechanism, our group has revealed that the binding of autoantibodies to antigens expressed in the keratinocytes triggers internalization of the antigenic targets in a process called macropinocytosis; resulting in a weakened dermal-epidermal adhesion and eventual subepithelial blistering (Hiroyasu et al., 2013, Am J Pathol - https://pubmed.ncbi.nlm.nih.gov/23337823/) (Figure 2).This has been demonstrated using live cell imaging techniques (https://www.youtube.com/watch?v=IYRKMPbSqKM), and was a previously unknown mechanism for bullous pemphigoid.
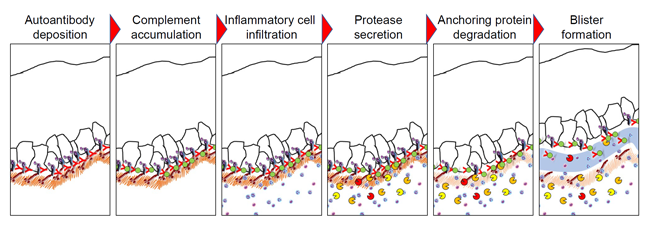
Figure 1: Complement and protease-mediated blistering in pemphigoid diseases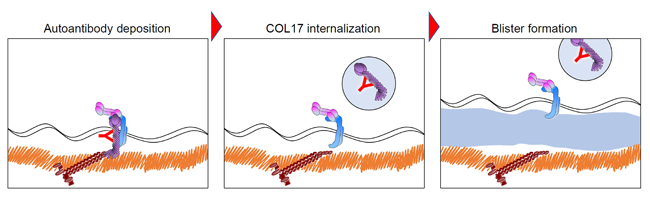
Figure 2: Blister formation due to the internalization of COL17, the autoantigen in bullous pemphigoidIn line with this study, we are currently focusing on the induction of cytokine release from epidermal keratinocytes by bullous pemphigoid IgG (https://kaken.nii.ac.jp/en/grant/KAKENHI-PROJECT-18K08305/). Through these studies, we aim to further clarify the processes occurring immediately after the onset of pemphigoid and identify potential targets for therapy in those stages.
-
Pathological functions of granzymes in pemphigoid
Granzymes are a type of serine proteases released from various types of the cells. Human granzymes are designated as Granzyme A, B, H, and K. Originally, these are known to be released together with perforin from cytotoxic T cells and natural killer cells during infection, being internalized into target cells through perforin-mediated pores to initiate apoptosis. However, in the recent years, it has been revealed that granzymes are released from various cell types including mast cells, basophils, and macrophages without concomitant perforin release to mediate non-cytotoxic, extracellular functions (Figure 3) ((Hiroyasu et al., 2021, J Dermatol Sci (https://pubmed.ncbi.nlm.nih.gov/34772583/)).
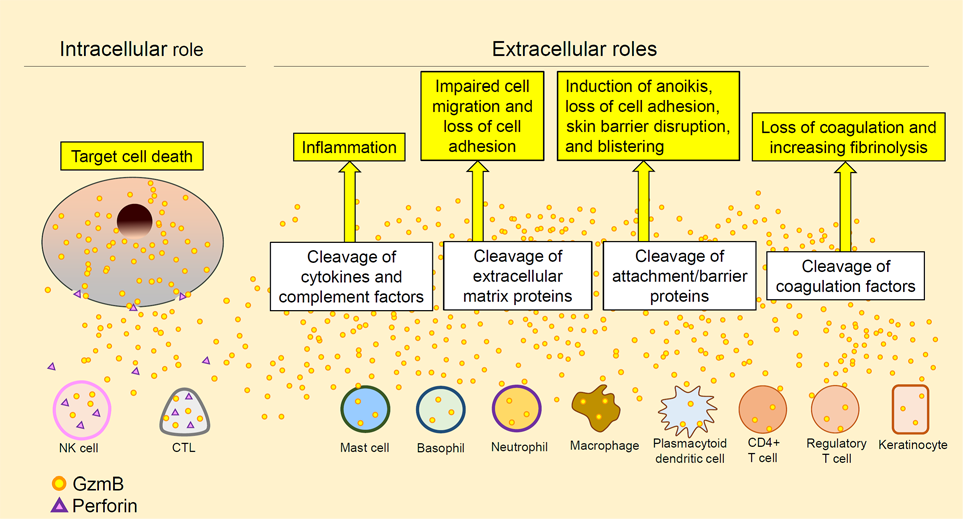
Figure 3. Intracellular and extracellular functions of granzyme B (excerpt from Hiroyasu et al., 2021, J Dermatol Sci)To test if these proteases can be a potential therapeutic target for the pemphigoid, we investigated the functions of granzyme B in the diseases in collaboration with Prof. David Granville’s lab at the University of British Columbia (Hiroyasu et al., 2021, Nat Commun (https://pubmed.ncbi.nlm.nih.gov/33436591/). Using multiple in vivo models and human samples, granzyme B released from mast cells and basophils has been shown to induce proteolytic degradation of key anchoring proteins at the DEJ and mediate IL-8 secretion from keratinocytes. Topical application of its novel small molecule inhibitor demonstrated improvement of blistering in murine models (Figure 4).
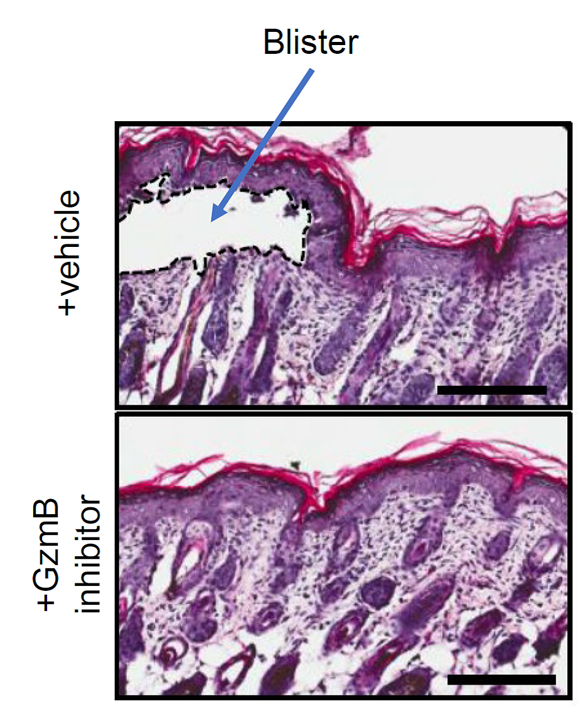
Figure 4. Topical application of a granzyme B (GzmB) inhibitor decreased severity in bullous pemphigoid mice (modified from Hiroyasu et al, 2021, Nat Commun).Currently, as an extension of this study, we are investigating the functions of granzymes in pruritus associated with pemphigoid disorders (https://kaken.nii.ac.jp/en/grant/KAKENHI-PROJECT-21K08305/). We are also conducting research to elucidate the mechanisms by which scratching exacerbates skin eruptions in pemphigoid diseases aiming to develop novel therapeutic modality through controlling itch in the diseases.
-
Other projects
In addition, our group is conducting functional analysis of granzyme B in vitiligo, and epidemiological research on pemphigoid diseases.
Positions Available!
Our group is currently looking for masters and doctoral graduate students who are willing to study with our lab. If you are interested in conducting research for autoimmune skin diseases, skin immunity, proteases, and cell adhesion factors, please contact Dr. Sho Hiroyasu (sho.hiroyasu@omu.ac.jp).
At the current moment, we are not recruiting postdoc fellows, international students, or laboratory technicians due to limited funding. However, if there are any fellowship grants that you wish to apply for in conjunction with our team, please feel free to contact our lab.
Lab Members:
| Primary Investigator | Sho Hiroyasu, MD, PhD (sho.hiroyasu@omu.ac.jp) google scholar : Sho Hiroyasu |
|---|---|
| Researcher/Lab Manager | Aoi Hiroyasu, MS |
| Postdoc | Bolla Beata Szilvia, PhD |
| PhD Student | Daisuke Matsumoto, MD |
| PhD Student | Hiroto Kuwabiraki, MD |
| PhD Student | Dieo Siwei, BS |
| PhD Student | Shingo Santo, MD |
