Research News
Oct 14, 2022
Smart materials: metal cations-recognizable thermoresponsive polymers
Osaka Metropolitan University scientists developed a novel polymer, the thermoresponsiveness of which can easily be regulated by changing the type and mixing ratio of ionic species.
Osaka, Japan – Often referred to as smart materials, temperature-responsive or thermoresponsive polymers are gaining attention for their ability to respond to external temperature changes, allowing for an extensive range of applications. Making this smart material even smarter by improving the flexibility of its response to temperature, Osaka Metropolitan University scientists have developed a novel polymer, the thermoresponsiveness of which can easily be regulated by changing the type and mixing ratio of ionic species. Their findings were published in Macromolecules.
Polymers that exhibit temperature-related change in their physicochemical properties are called thermoresponsive polymers. They include two types: polymers with lower critical solution temperatures (LCST) and polymers with upper critical solution temperatures (UCST). Above a certain temperature, the former are insoluble, whereas the latter are soluble. In LCST-type thermoresponsive polymers, as the temperature increases, the polymer–solvent interaction decreases and the polymer–polymer interaction becomes dominant, leading to precipitation of polymers from the solvent. Conversely, in UCST-type thermoresponsive polymers, as the temperature increases, the polymer–polymer interaction decreases and the polymer–solvent interaction becomes dominant, resulting in dissolution. This indicates the affinity between the polymer and the solvent as an important factor in most thermoresponsive polymers.
The novel thermoresponsive polymer was created by adding ions to polymers and aqueous solutions. Its thermoresponsiveness can easily be controlled by changing the type and mixing ratio of ionic species.
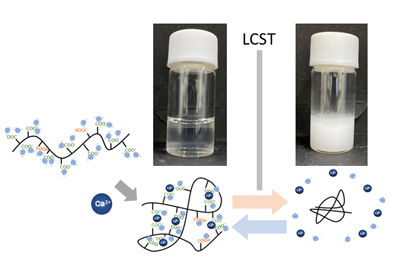
Conventionally, the polymer–solvent interaction is used to regulate thermoresponsiveness in designing thermoresponsive polymers. However, attention has recently focused on a new technique that regulates thermoresponsiveness by adding a third component. This technique often uses organic solvents, but to develop materials such as those for drug delivery systems, it is necessary that water, which is harmless to the human body, be used as the solvent.
The research team led by Professor Atsushi Harada from the Graduate School of Engineering at Osaka Metropolitan University used water as the solvent and developed an LCST-type thermoresponsive polymer by adding alkaline earth metal ions—which are divalent cations—to polymers and aqueous solutions. They succeeded in regulating thermoresponsive properties, simply by changing the species and mixing ratio of the ions. This is different from the conventional technique that can only regulate thermoresponsiveness by changing the structure of the polymers.
“We have developed a novel polymer that exhibits thermoresponsiveness in the presence of certain ions,” concluded Professor Harada. “We expect that it will be applied as an analytical reagent for metal ion-sensing devices and as a material for drug delivery systems.”
Funding
This work was partially supported by the Leading Initiative for Excellent Young Researchers, MEXT, Japan.
Paper Information
Journal: Macromolecules
Title: Thermoresponsiveness of Carboxylated Polyallylamines Induced by Divalent Counterions as Ionic Effectors
DOI: 10.1021/acs.macromol.2c00795
Author: Junya Emoto, Yukiya Kitayama, Atsushi Harada
Publication date: July 28, 2022
Macromolecules (ACS Publications Website)
Contribution to SDGs

9: Industry, Innovation and Infrastructure
Contact
Graduate School of Engineering
Prof. Atsushi HARADA
E-mail: atsushi_harada[at]omu.ac.jp
*Please change [at] to @.
SDGs

