Research News
Dec 14, 2022
Shedding light on photosynthesis at sea
Osaka Metropolitan University scientists use cryogenic electron microscopy to investigate how the marine green macroalga Codium fragile photosynthesizes
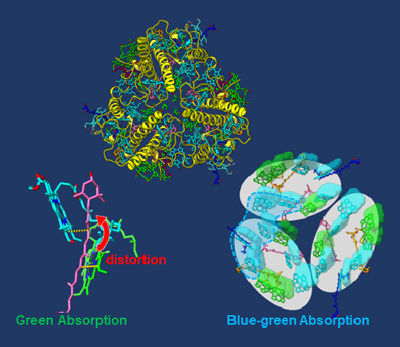
Using cryogenic electron microscopy, Osaka Metropolitan University scientists found that siphonaxanthin of C. fragile is greatly distorted and forms hydrogen bonds with the surrounding protein at two locations. The researchers also clarified several chlorophyll molecule substitution sites. When the substitution occurs, the adjacent region of chlorophyll b clusters becomes wider, enabling more efficient absorption of blue-green light. These structural features allow C. fragile to better utilize blue-green light for photosynthesis.
Plants that live on land, such as spinach, grow by using sunlight to perform photosynthesis. How, then, do algae photosynthesize in the deep sea, an environment where only a little light reaches them?
Land plants mainly absorb red and blue light from the sun and use it for photosynthesis. However, only weak blue-green light reaches the ocean floor. Therefore, macroalgae growing in the ocean have developed a protein, a so-called photosynthetic antenna, that efficiently utilizes this blue-green light. The photosynthetic antenna of marine macroalgae is very similar to that of land plants but differs in the structure of the pigments bound to it. Land plants have two types of pigments bound to their photosynthetic antennae, namely carotenoids and chlorophylls. In the marine green macroalga Codium fragile, the major carotenoids are substituted with siphonaxanthin while some chlorophyll a molecules are replaced by chlorophyll b molecules. Siphonaxanthin and chlorophyll b are known to contribute to increased absorption of green light and blue-green light, respectively, but the mechanism has not yet been fully understood.
Responding to this gap, a research team led by Associate Professor Ritsuko Fujii, from the Research Center for Artificial Photosynthesis (ReCAP) at Osaka Metropolitan University, and graduate student Soichiro Seki, from the Graduate School of Science at Osaka City University, used cryogenic electron microscopy to investigate the structures and binding environments of pigments bound to the photosynthetic antenna of C. fragile. The results allow for the elucidation of the molecular mechanism by which blue-green light—the only light available in deep seawater—is efficiently utilized for photosynthesis. Their findings were published in BBA Advances on November 11, 2022.
High-resolution analysis by cryogenic electron microscopy showed that siphonaxanthin in C. fragile is greatly distorted and forms hydrogen bonds with the surrounding protein at two locations. This structural feature is deemed a key factor in siphonaxanthin’s ability to absorb green light. Additionally, the researchers successfully detected the difference between chlorophyll a and chlorophyll b, and they clarified several chlorophyll molecule substitution sites. When the substitution occurs, the adjacent region of chlorophyll b clusters becomes wider, enabling better absorption of blue-green light. In other words, the team was able to obtain information on the pigment coordinates, contributing to a better understanding of the mechanism of more efficient photosynthesis.
“I think increasing the utilization of photosynthesis simply by changing the pigment structure would be a cost-effective strategy,” explained Professor Fujii. “Learning such survival strategies of organisms would lead to improved use of sunlight and the development of renewable energy sources for human beings.”
Funding
This study was funded by the grant-in-aid of the Sasakura Enviro-Science Foundation, the OCU Strategic Research Grant for Basic Researches, and the JST-CREST under grant number JPMJCR20E1, SUNTORY Foundation for Life Sciences Bioorganic Research Institute for a scholarship (FY2020-2023). This work was performed in part under the Collaborative Research Program of the Institute for Protein Research, Osaka University (CR-21-02), and the joint research program of the Artificial Photosynthesis, Osaka City University (FY2018).
Paper Information
Journal: BBA Advances
Title: Structural insights into blue-green light utilization by marine green algal light harvesting complex II at 2.78 Å
Author: Soichiro Seki, Tetsuko Nakaniwa, Pablo Castro-Hartmann, Kasim Sader, Akihiro Kawamoto, Hideaki Tanaka, Pu Qian, Genji Kurisu, Ritsuko Fujii
DOI: 10.1016/j.bbadva.2022.100064
Publication date: November 11, 2022
https://doi.org/10.1016/j.bbadva.2022.100064
Contact
Graduate School of Science
Ritsuko Fujii
E-mail: ritsuko[at]omu.ac.jp
*Please change [at] to @.
SDGs

